Liquid Biopsy for Cancer, Pros and Cons
January 26, 2021 | Terry Sharrer
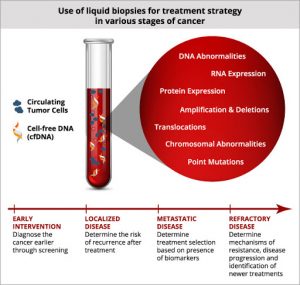
Liquid biopsy
“. . . liquid biopsy entered a new era in August with FDA’s approval of two tests that use next-generation sequencing (NGS) to target many genes in advanced cancer patients, including mutations for which there are targeted drugs, and to profile any solid tumor. These new tests are Guardant Health’s Guardant 360 CDx assay, and FoundationOne Liquid CDx, marketed by Foundation Medicine. . . . [Both] allow oncologists to focus care on the molecule of origin rather than the cell type . . .” MORE
Image Credit: healio.com


